Rutgers: Douglas E. Eveleigh (1933-2019)
- Ray Sullivan

- Jan 28, 2020
- 29 min read
Updated: Feb 2, 2025
Douglas Eveleigh, an intellectual giant passed away on December 30, 2019, a victim of glioblastoma. He was Distinguished Professor Emeritus in the Dept of Biochemistry & Microbiology, Rutgers University. In 2001, he was invested as the first holder of the Douglas and Linda Eveleigh and Dennis and Linda Fenton Endowed Chair in Applied Microbiology. Douglas Eveleigh was a leading researcher and teacher at Rutgers for 45 years, mentoring 31 PhD and 12 MS students. He was a tireless supporter of the Theobold Smith Society, as a past president of the society and an Honorary Selman Waksman Award speaker.
While visiting Doug this past September, I asked Doug about his own professional legacy. He thought a moment and concluded it was his publications. The following post aims to record those publications and tell a short story of Doug’s professional life. For a more succinct (and interesting) obituary see the one written by Max Häggblom and Joan Bennett.

Douglas Eveleigh was born December 6, 1933 and raised just south of London. He was a naturalized American and never lost use of the Queen’s English. During World War II, young Doug was evacuated (along with 3.5 million others) to areas in Britain thought to be less at risk for German aerial bombing. Later in life, to amuse his Rutgers colleagues, he would occasionally wear his father’s bowler hat as a nod to his English roots.
Doug received his B.Sc. from the University of London in 1956 and went on to earn his PhD from the Department of Botany, University College, Exeter (UK) in 1958. At Exeter, he studied fungi attacking painted surfaces under the direction of Professor John Caldwell, Director of the Hatherly Biological Laboratories, legendary mycologist Dr Geoffrey C. Ainsworth, Director of the Commonwealth Mycological Institute at Kew (Ainsworth & Bisby’s Dictionary of the Fungi) and Dr Sydney A.J. Tarr, Reader in Plant Pathology (Eveleigh, 1961a, Eveleigh, 1961b, Eveleigh, 1961).
Opportunities were probably very limited in post-war Britain, so he looked elsewhere and landed his first post-doc with the Atlantic Regional Laboratory, National Research Council of Canada, Halifax, Nova Scotia. From 1959 to 1961, he studied slimes obtained from paper mills in Eastern Canada which started him on an intellectual journey with cellulose that lasted a lifetime. He investigated cellulolytic fungal and bacterial thermophiles (Eveleigh and Brewer, 1963), psychrophiles (Eveleigh and Brewer, 1964a), their nutritional requirements (Eveleigh and Brewer, 1964b) and their ecological interrelationships (Eveleigh and Brewer, 1965).
His next post-doc in 1962 was with Elwyn Reese and Mary Mandels at the US Army Natick Laboratories, in Natick, MA (Eveleigh et al., 1964). Reese and Mandels had isolated a mold, Trichoderma viride, from rotting military clothing and tents from Solomon Islands in the South Pacific during World War II. The fungus stimulated research on how insoluble polymeric substrates (such as the cotton, linen or hemp canvas tents) are utilized by microorganisms and whether secretion of extracellular cellulase enzymes were involved. This research started Eveleigh’s life-long study of enzymes that could be used for industrial purposes such as converting cellulosic materials into glucose to produce alcohol. More importantly, while at Natick he met Linda - they were happily married for 57 years. At the time, Linda was secretary to Harvard Professor Bernie Davis, a pioneer in microbial physiology and metabolism. Davis was coincidentally the first recipient of the Theobold Smith Society’s Waksman Honorary Lectureship in 1954.

During a post-doc at University of Wisconsin, Madison, their first son Chris was born. He then landed a second job with the National Research Council of Canada, this time at the Prairie Regional Laboratory, Saskatoon, Saskatchewan, Canada. There, he solidified his expertise with cellulase and other polysaccharases (Sietsma et al., 1967, Eveleigh, 1967, Gamborg and Eveleigh, 1968, Eveleigh et al., 1968, Sietsma et al., 1968, Eveleigh and Perlin, 1969, Sietsma et al., 1969, Gorin et al., 1969, Eveleigh and Child, 1969, Gorin and Eveleigh, 1970, Child et al., 1973a, Child et al., 1973b). He used the enzymes to make protoplasts for plant cell hybridization which was then a relatively new thing and he scored a Nature paper (Keller et al., 1970). Rob, his second son, was born in Saskatoon in 1966.
During summers in Saskatoon, Linda Eveleigh’s New England roots beckoned the family of four back for vacations on the island of Nantucket off the Massachusetts coast. While he and his family relaxed, he also took it as an opportunity for research and studied how various fungal species attacked cedar shingles on Nantucket’s houses (Eveleigh, 1970). Nantucket research would continue throughout his very productive scientific career. Thirty years later he was still publishing research from Nantucket (Cronin et al., 2000, Benson et al., 2004). It is said, if you love your job you never have to work. Doug supposed you may as well bring it on vacation too.
In 1970, Doug landed an associate professorship in the Department of Biochemistry and Microbiology, College of Agriculture and Environmental Science, Rutgers University in New Brunswick, N.J. It was here he would spend the rest of his dynamic career and retire in 2014 as Distinguished Professor.

Doug was a leader, not a manager. All one had to do was walk into his office to realize this: it was the most disorganized grabastic center of entropy on the campus. Books, papers, old instruments, mementos, flotsam and jetsam was piled up everywhere. If you went to his office for advice (which many people often did) he’d have to move a pile of papers and books to uncover a chair or easier still, just leave his office and rush down the hall to the conference room where there were plenty of chairs. Every evening, he would leave Lipman Hall toting half a library of material for further work at home and lug it all back in the morning.
He led by influence, not power. None of his students felt like subordinates; we were his followers. He inspired, motivated and enabled his lab (and beyond) to achieve scientific success, but controlled nothing. He never spoke of tasks, but always had vision, purpose and aspiration. He focused on people, trusted us and challenged us to work hard. Working with Doug was never a chore, it was very enjoyable. It was fun.
What distinguished him was his ability to spot interesting scientific developments and quickly act on them. He read relentlessly and wrote prolifically. He talked to colleagues all over the world and invited them to Rutgers for talks. He got out of the office often to give talks and attend conferences. He took sabbaticals whenever possible. Although there were overall career research themes like polysaccharases, he would not stick with a particular organism or approach for long. He was constantly reinventing his research.
Like most professors, Doug spent most of his time publishing and writing grant proposals. There were only so many hours in a day and he did not waste a lot of time preparing his course lectures. After all, he knew the stuff cold; why organize it too much? He had no time to waste. I had a 3.9 GPA when I took my qualifiers and was very proud of it. Doug burst my bubble and told me, “You wasted too much time studying course work. You should have been doing more research!” He was right.

Sweeteners When Doug moved to Rutgers in 1970, US FDA banned the artificial sweetener sodium cyclamate (saccharin) presenting an opportunity to find safer alternatives. Eveleigh teamed up with Harald Linke at NYU to investigate methods to convert the bitter citrus peel compounds naringenin and neohesperidin into naringin dihydrochalcone and neohesperidin dihydrochalcone which are 300-1800 times sweeter than sugar (Linke and Eveleigh, 1975b, Linke and Eveleigh, 1975a, Natarajan et al., 1976). Their method resulted in Doug’s first patent (Linke and Eveleigh, 1978). Doug did not continue working with the compounds, but interest in them has since taken off. Neohesperidin dihydrochalcone, in particular, is used as a sweetener in a variety of products in the European Union, but not the US.

Nitrogen fixation Nantucket vacations again proved favorable for the Doug’s well-prepared mind. He became interested in how plants growing on the nutrient-poor soils of Nantucket (and also Jersey Shore) obtained their nitrogen. He found nitrogen fixation occurred via symbiotic plant bacterial endophytes in the root nodules of bayberry (but not bearberry) growing on the dunes (Morris et al., 1974, Tiffney Jr et al., 1978). Subsequent electron microscopic studies of the nodules by Doug’s graduate student, Dave Benson, tentatively identified the actinorhizal endophytes as a Frankia sp. (Benson and Eveleigh, 1979a, Benson et al., 1979, Benson and Eveleigh, 1979b, Eveleigh, 1992). From the University of Connecticut, Professor Dave Benson would go on to build a career around Frankia
research. Coincidentally, Dave’s undergraduate son, now Dr Micah Benson, also got a research start on Nantucket. Under the tutelage of his dad and Doug in the summer of 2003, Micah investigated bacteria associated with deer ticks collected on Nantucket (Benson et al., 2004).
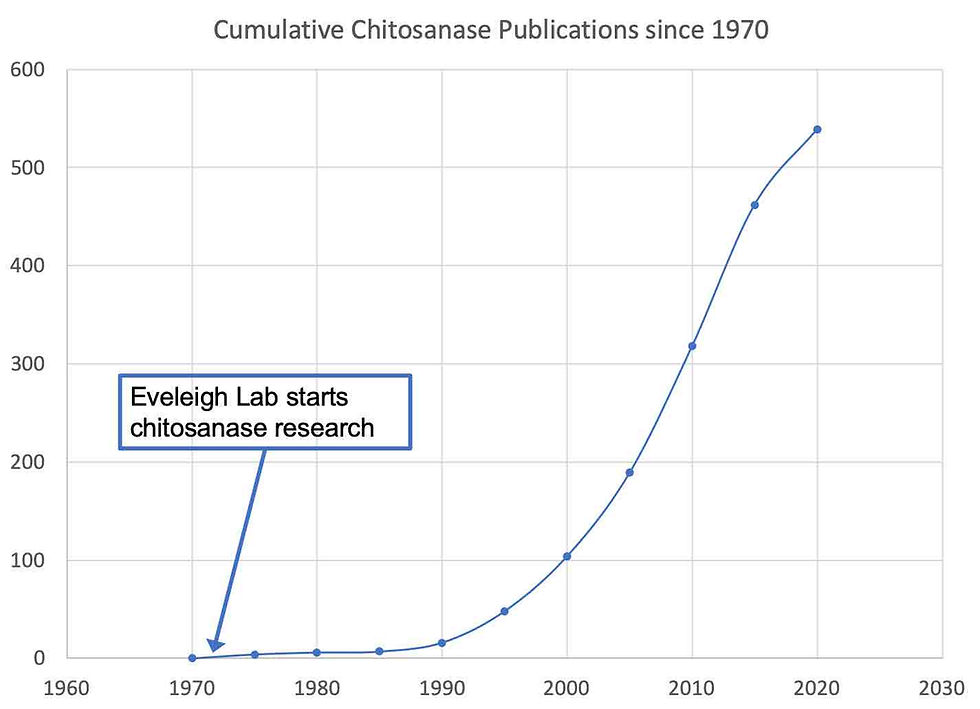
Chitosanase Professor Eveleigh’s main career research effort involved microbial enzymes that rip apart plant and fungal polysaccharides. He was already very experienced with polysaccharases from his post-doc days at Natick and Saskatoon. His first polysaccharase projects at Rutgers were with chitosanases, the work probably driven by US Army funding opportunities. Chitosan is a major component of Mucor sp. cell walls and mucormycosis afflicts burn and other wound patients. It is difficult to treat and often lethal. The Army was looking at chitosanases as a possible treatment since it would conceivably rip apart the Mucor cell wall. Doug and his graduate students, Dick Monaghan, Dennis Fenton, Brian Davis were the first to investigate chitosanases from various organisms (Monaghan et al., 1973, Fenton et al., 1978, Fenton and Eveleigh, 1981, Davis and Eveleigh, 1984). Their efforts spawned a lively and productive field of research that has not slowed.

Trichoderma As was mentioned earlier, Professor Eveleigh became well known for Trichoderma cellulase research ever since his post-doc at Natick with Reese and Mandels. The oil embargo/energy crisis in the 1970s stimulated the idea that cellulases could have an impact by conceivably producing fuels from renewable lignocellulosic biomass economically. Over a fifteen-year period, his lab worked to produce high-yielding cellulase secreting Trichoderma mutants, especially Trichoderma reesei strain RUT-C30. The strain remains the prototype cellulase hyperproducer (30g/L) available to the public domain and is the basis of many commercial strains. It is mentioned in over one hundred cellulase patents involving bioenergy – plant waste to ethanol conversion, as well as other industrial applications such as stone washing of denim and detergent additives. While government funding for cellulase/biomass conversion to energy projects have yet to achieve commercialization goals, it has produced many PhDs that have sharpened the US competitive edge in the global economy. Doug provided six of them: Gene Frein, Steve Picataggio, Kathy Holland, Stephanie Zitomer, Sawsan M. El-Gogary, and Doty Klein (Eveleigh, 1981, Frein et al., 1982, Stokes et al., 1983a, Picataggio et al., 1983, Eveleigh, 1983, Bartley et al., 1984, Flavell et al., 1985, Eveleigh, 1985, Zitomer and Eveleigh, 1987, Eveleigh, 1987b, el-Gogary et al., 1989, Bisaria et al., 1989, Kubicek et al., 1990, Goyal et al., 1991, Eveleigh, 1994, Klein and Eveleigh, 1998). More than any other research area Doug kick-started, Trichoderma research has exploded.
Doug worked closely on Trichoderma cellulases with Bland Montenecourt over a ten year period; she was another successful Eveleigh protégé (Montenecourt and Eveleigh, 1977b, Montenecourt and Eveleigh, 1977a, Montenecourt et al., 1978, Eveleigh and Montenecourt, 1979, Montenecourt and Eveleigh, 1979, Montenecourt et al., 1981, Frein et al., 1982, Ghosh et al., 1982, Schimenti et al., 1983, Picataggio et al., 1983, Ghosh et al., 1984, Kelleher et al., 1987, Eveleigh, 1987a). Doug brought Bland aboard the Department of Biochemistry and Microbiology in 1976 as soft money faculty. She left the department as an assistant research professor in 1981, accepting a faculty position at Lehigh University as an associate professor and was promoted to full professor in 1985. Tragically, Professor Montenecourt was felled by cancer at the young age of 45.

Zymomonas While Trichoderma was now more efficient at breaking down cellulose, it did not ferment sugars to ethanol. A recombinant organism was needed and Doug briefly looked at using alcohol fermenting yeasts as vehicles for his Trichoderma enzymes (Burden and Eveleigh, 1990). Instead of yeasts, he chose to continue with much more genetically tractable bacteria instead. Eveleigh was an early research leader using Zymomonas mobilis for biofuel production. The bug is a natural ethanol secretor with many desirable characteristics potentially making it a candidate to directly convert lignocellulose to ethanol through metabolic engineering. He knew that to directly convert cellulose to ethanol, Z. mobilis would need to be easily genetically modifiable, need an expanded substrate range (it naturally grows only on glucose, fructose and sucrose) and would need to secrete cellulase enzymes (endoglucanase, exoglucanase and β-glucosidase). From 1980 to 1995, his lab made progress in bioengineering all of these (Dally et al., 1982, Stokes et al., 1983b, Eveleigh et al., 1983, Buchholz and Eveleigh, 1986, Lejeune et al., 1986, Buchholz et al., 1987, Lejeune et al., 1988, Buchholz et al., 1988, Yablonsky et al., 1988b, Buchholz et al., 1989, Buchholz and Eveleigh, 1990, O'Mullan et al., 1991, O'Mullan et al., 1992, Lam et al., 1993, O'Mullan et al., 1995). Twenty-five years later, work is still ongoing at the US National Renewable Energy Laboratory (NREL) as well as in China to engineer Zymomonas for cellulosic ethanol production. Many PhDs were minted with the NREL money and Doug used it to shepherd four of them through: Ellen Dally, Steve Buchholz, Pat O’Mullan and Cuong Lam.
Thermophiles Being an industrial microbiologist, Doug knew thermostable enzymes offered potential advantages in the hydrolysis of cellulosic materials. Microbial producers of thermostable cellulases are very efficient at degrading lignocellulosic biomass. Enzymes from thermophiles are generally more stable than those from mesophilic organisms. Industrially, lignocellulose normally needs thermal pretreatment and in using a thermostable enzyme in a high temperature reactor, cooling energy requirements are less. The increased solubility of heat-treated cellulose combined with high temperature enzymes results in higher enzyme velocity so less enzyme and hydrolysis time is required. There is also reduced risk of contamination by mesophiles and psychrophiles in a high temperature industrial process. High temperature also facilitates recovery of ethanol. To find the enzymes necessary, he looked to the thermophilic actinomycetes (Hunter-Cevera and Eveleigh, 1990, Nash et al., 1992, Hunter-Cevera et al., 1996).

Microbispora bispora is an obligate thermophile growing only between 50-65°C and found on compost and other decaying material. Interestingly, Professor Hubert Lechevalier, a friend and colleague of Doug’s at Rutgers (and one of Selman Waksman’s students) changed the organism’s name from Thermopolyspora bispora to Microbispora bispora in 1965 since he was felt that thermophily should not be used as a criterion to differentiate between genera of Actinomycetes. The organism was again renamed in 1996 and is now Thermobispora bispora. Clarence Waldron, a student of Eveleigh’s studying composting in the Environmental Science Department at Rutgers, isolated the lab’s particular bacterial strain. It produced high yielding extracellular cellulase and started a 10 year project to characterize and utilize the bacterium’s thermostable enzymes (Waldron et al., 1983, Bartley et al., 1984, Waldron et al., 1986, Eveleigh et al., 1986, Waldron and Eveleigh, 1986, Yablonsky et al., 1988a, Yablonsky et al., 1989, Elliston et al., 1991, Hu et al., 1992, Wright et al., 1992, Goyal and Eveleigh, 1992, Hu et al., 1993, Eveleigh et al., 1995, Goyal and Eveleigh, 1996). M. bispora was one of the only Eveleigh projects that did not gain much traction with the wider scientific community and use of the bacterium’s cellulases were never used on an industrial scale for biomass to energy conversion. It takes a little longer for the microbe to grow – two days instead of overnight, but the main unattractiveness was its high GC content which made cloning and sequencing the microbe difficult. Importantly, the research funded an MS degree for Yongmei Wu and Sue Kahrs and five PhDs: Tim Bartley, Keith Elliston, Anil Goyal, Rick Wright and Chip Yablonsky.

Thermotoga Professor Eveleigh became interested in Thermotoga maritima and Thermotoga neapolitana shortly after his colleagues Holger Jannasch, Karl Stetter and Robert Huber discovered them. Thermotoga sp. are among the most ancient and thermophilic bacteria known. They grow at temperatures up to 90°C and produce a wide range of thermostable polysaccharases which Doug’s lab was among the first to characterize. The Eveleigh lab found T. neapolitana’s polysaccharases (endoglucanases, β-glucosidases, β-1,4-xylanase, 1,4-β-D-glucan glucohydrolase) had high specific activity and are very stable to extreme temperatures (>95°C), optimal characteristics for industrial applications including converting plant biomass to ethanol (Bok et al., 1994, Bok et al., 1998, Yernool et al., 2000a). The lab used directed evolution techniques to mutate T. neapolitana’s 1,4-β-D-glucan glucohydrolase in order to increase its activity by 31% (McCarthy et al., 2003, McCarthy et al., 2004). It is now generally accepted that production of plentiful amounts of simple sugars from highly complex lignocellulose in an economically viable manner requires thermostable cellulose and hemicellulose systems such as those from Thermotoga sp. (Eveleigh, 2003).

Leguminous foods such as beans and peanuts contain α-1,6-galactosides that remain undigested until they pass into the large intestine where they are broken down by the intestinal microbiota to yield considerable amounts of gas (methane, carbon dioxide and hydrogen) causing gastrointestinal as well as societal distress. Beano, a human dietary supplement containing an α-1,6-galactosidase is used to reduce gas in the digestive tract improving digestion and reducing bloating, discomfort, and flatulence caused by the gas. A thermostable T. neapolitana “beano” was characterized and patented for industrial applications such as high temperature conversion of soy bean molasses to more readily digestible carbohydrates (King et al., 1998, Yernool et al., 2000b). The Thermotoga research funded PhDs for Jin Duck Bok, Dinesh Yernool, Flip McCarthy, Gavin Swiatek and an MS for Julie Antillon.

Professor Jim Macmillan was Department Chair in 1970 and hired Doug. They were close personal friends ever since. Jim was just as eclectic in his research interests as Doug so funding opportunities sometimes presented openings to share research as well as graduate students. Tom Jeffries and Frank Riske were two of Jim’s students who worked on polysaccharide degrading enzymes with Doug (Jeffries et al., 1977, Riske et al., 1986, Riske et al., 1987, Riske et al., 1990). Frank Ritacco was Doug’s last graduate student. Frank had studied under Jim Macmillan and graduated in 1996 with an MS. He then went to work for Wyeth Research. Several years later, Doug found a way for Frank to continue graduate studies while working full time at Wyeth. Frank earned his PhD in 2007 (Ritacco and Eveleigh, 2008).
Starting in 2001, Professor Eveleigh had a long research collaboration with Professor Hunsa Punnapayak from the Chulalongkorn University, Bangkok, Thailand. Professor Punnapayak’s interests in biomass and crop residue transformation and utilization using fermentation and fungal enzymes meshed nicely with Doug’s research efforts. Together, they studied how the yeast Aureobasidium pullulans produced pullulan, a polysaccharide polymer used in the production of edible films (Prasongsuk et al., 2005, Prasongsuk et al., 2007, Manitchotpisit et al., 2009, Manitchotpisit et al., 2011, Bankeeree et al., 2014, Yanwisetpakdee et al., 2016). They also studied polysaccharase enzymes produced by Fusarium solani, Aspergillus flavus, Phanerochaete sordida, Ceriporia cystidiata, Macrohyporia dictyopora as well as from the bacterium Lysobacter enzymogenes (Nimchua et al., 2008, Danmek et al., 2011, Permpornsakul et al., 2016, Saraihom et al., 2016, Permpornsakula et al., 2016). Doug trained several of Hunsa’s students in Lipman Hall starting with Sehanat Prasongsuk and continuing with Thidarat Nimchua, Pennapa Manitchotpisit, Khanchai Denmek, Wichanee Bankeeree, Benjawan Yanwisetpakdee, Polada Ppermpornsakul and Saraihom Siraprapa. After Doug retired in 2014, Jim White and Don Kobayashi in Plant Biology helped Doug in these efforts.
In another international collaboration, Professor Aline Aparecida Pizzirani Kleiner from the Dept. of Genetics, University of São Paulo, Brazil sent her graduate student André Lima to learn molecular techniques with Doug for a year. Lima constructed a GFP- β-glucosidase fusion protein using GFP fluorescence as a quick screen surrogate to monitor the directed evolution of the protein (Lima et al., 2009).
Doug would often say, “If it’s not published, it didn’t happen.” He wrote reviews of books about soil enzymes (Eveleigh, 1979), nitrogen fixing organisms (Eveleigh, 1992) and mushrooms (Eveleigh, 2012). Obituaries were important to him and is a good reason for this one. He wrote about he lives of his friends and colleagues: Bland Montenecourt (Gottlund and Eveleigh, 1988), Mary Mandels (Allen et al., 2009), the industrial microbiologist Gerhard Haas (Eveleigh and Bennett, 2013), and H. Boyd Woodruff, Selman Waksman’s student who discovered actinomycin (Bennett et al., 2017).
Doug published a couple of post-humous papers written by Mary Mandels. One, a methods paper, Measurement of saccharifying cellulase was originally published in 1976. Shortly after Mandel’s death Doug re-published it to remind his colleagues of the origin of the method, its significance and simplicity as well as the prominence of the author, Mandels (Eveleigh et al., 2009). The second paper is much more remarkable. In her commentary Reflections on the United States military 1941-1987, Mandels reflects on her 46-year career as an Army research scientist. She pulls no punches and lays out a persuasive argument of what ails Army R&D, the military and its associated industrial base in general as well as the nation as a whole. As a retired naval officer and now as an Army research scientist myself, I was very appreciative of her analysis and think her remedies still relevant 10 years after publication. I have shared the paper with my Army colleagues (Mandels and Eveleigh, 2009).

The first time I met Professor Eveleigh was in his lab section of Microbial Ecology. Richard Bartha taught the lecture in a very organized way: I learned a lot. The labs were much different – impromptu – and a hell of a lot of fun. Doug and his grad student, Jin Duck Bok sort of winged it. The lab reports were not even really graded: if you turned one in you got credit. The most memorable exercise was a field trip to a salt marsh to gather material for a Winogradsky column. Doug loved them and wrote an article about them (Eveleigh and Davis, 1996). Tamar Barkay gave him a whimsical Winogradsky column as a gift a few months before he died.

Doug was enthusiastic about the history of microbiology and was honored by being chosen to be on a committee of senior American Society for Microbiology (ASM) members who prepared a list of the 26 most consequential discoveries in microbiology for the centennial exhibit at the ASM General Meeting in 1999 (Baker et al., 1999). He was proud of the scientific achievements of his adopted country, the state of New Jersey and scientific acumen of the first president of the United States, crediting the discovery of methane to Washington. Eveleigh and Ted Chase wrote, “George Washington and Thomas Paine demonstrated in November 1783 on the Millstone River at Rocky Hill, NJ that what was called setting the river on fire was setting on fire was setting the inflammable air that arose out of the mud.” (Eveleigh and Chase, 1997). Unfortunately, Washington and Paine’s discovery did not make the list of 26 most consequential discoveries in microbiology.

He was especially proud of Rutgers place in history. Doug was keenly interested in the rancorous legal battle between Selman Waksman and his graduate student Albert Schatz over who deserved credit for the discovery of Streptomycin. Doug was influential in establishing the Waksman Museum in the basement of Martin Hall on Cook Campus where Waksman, Schatz and Elizabeth Bugie made the streptomycin discovery that reaped the Nobel Prize for Waksman. He campaigned hard for Streptomyces griseus, producer of streptomycin, to become the New Jersey State Microbe (Bennett and Eveleigh, 2010) and lived to see it happen. Towards the end of his career, Doug along with Max Häggblom, Jeff Boyd and Joan Bennett wrote several articles about antibiotics research at Rutgers (Eveleigh and Häggblom, 2013, Eveleigh et al., 2015, Eveleigh and Bennett, 2018, Häggblom et al., 2019).

Sheer fun! It was the fitting title of his 2014 retirement celebration. He would say it with a laugh almost. It was the way he approached science and the way he lived. He made Lipman Hall a fun place to be and because of this, Lipman Hall is hallowed ground for most all of Doug’s students. Douglas Eveleigh will be sorely missed.


PUBLICATIONS
1. Allen, F; Andreotti, R; Eveleigh, DE; Nystrom, J. (2009) Mary Elizabeth Hickox Mandels, 90, bioenergy leader. Biotechnology for Biofuels 2:22.
2. Baker, P; Benoit, R; Bibel, DJ; Brock, T; Chung, KT; Domer, J; Eveleigh, D; Faison, B; Isenberg, H; Jannasch, H; Lederberg, J; Schaechter, M; Schmidt, M; Silver, S; Summer, W; Whitt, D. (1999) Microbiology’s fifty most significant events during the past 125 years. ASM News 65:U7.
3. Bankeeree, W; Lotrakul, P; Prasongsuk, S; Chaiareekij, S; Eveleigh, DE; Kim, SW; Punnapayak, H. (2014) Effect of polyols on thermostability of xylanase from a tropical isolate of Aureobasidium pullulans and its application in prebleaching of rice straw pulp. Springerplus 3:37.
4. Bartley, TD; Murphy-Holland, K; Eveleigh, DE. (1984) A method for the detection and differentiation of cellulase components in polyacrylamide gels. Analytical Biochemistry 140:157-161.
5. Bennett, JW; Eveleigh, DE. (2010) State microbes. Microbe 5:412.
6. Bennett, JW; Eveleigh, D; Goodman, RM. (2017) H. Boyd Woodruff (1917–2017). Science 356:381.
7. Benson, DR; Eveleigh, DE. (1979a) Nitrogen-fixing homogenates of Myrica pensylvanica (Bayberry) non-legume root nodules. Soil Biology and Biochemistry 11:331-334.
8. Benson, DR; Eveleigh, DE. (1979b) Ultrastructure of the nitrogen-fixing symbiont of Myrica pensylvanica L.(Bayberry) root nodules. Botanical Gazette 140:S15-S21.
9. Benson, DR; Greenhut, VA; Eveleigh, DE. (1979) Scanning electron microscopy of the nitrogen-fixing actinorhiza of Myrica pensylvanica L. (bayberry). New Phytologist 83:755-760.
10. Benson, MJ; Gawronski, JD; Eveleigh, DE; Benson, DR. (2004) Intracellular symbionts and other bacteria associated with deer ticks (Ixodes scapularis) from Nantucket and Wellfleet, Cape Cod, Massachusetts. Applied and Environmental Microbiology 70:616-620.
11. Bisaria, VS; Mishra, S; Eveleigh, DE. (1989) Regulatory aspects of cellulase biosynthesis and secretion. Critical Reviews in Biotechnology 9:61-103.
12. Bok, JD; Yernool, DA; Eveleigh, DE. (1998) Purification, characterization, and molecular analysis of thermostable cellulases CelA and CelB from Thermotoga neapolitana. Applied and Environmental Microbiology 64:4774-4781.
13. Bok, JD; Goers, SK; Eveleigh, DE. (1994) Cellulase and Xylanase Systems of Thermotoga neapolitana, p. 54–65. In Himmel, ME; Baker, JO; Overend, RP (eds.), Enzymatic Conversion of Biomass for Fuels Production, American Chemical Society, Washington, DC.
14. Buchholz, SE; Dooley, MM; Eveleigh, DE. (1989) Growth of Zymomonas on lactose: gene cloning in combination with mutagenesis. Journal of Industrial Microbiology 4:19.
15. Buchholz, SE; Eveleigh, DE. (1986) Transfer of plasmids to an antibiotic-sensitive mutant of Zymomonas mobilis. Applied and Environmental Microbiology 52:366-370.
16. Buchholz, SE; Eveleigh, DE. (1990) Genetic modification of Zymomonas mobilis. Biotechnology Advances 8:547-581.
17. Buchholz, SE; Dooley, MM; Eveleigh, DE. (1987) Zymomonas — an alcoholic enigma. Trends in Biotechnology 5:199-204.
18. Buchholz, SE; O’Mullan, P; Eveleigh, DE. (1988) Growth of Zymomonas mobilis CP4 on mannitol. Applied Microbiology and Biotechnology 29:275-281.
19. Burden, DW; Eveleigh, DE. (1990) Yeasts - diverse substrates and products., p. 199–227. In Spencer, JFT; Spencer, DM (eds.), Yeast Technology, Springer-Verlag, Berlin.
20. Child, JJ; Eveleigh, DE; Sieben, AS. (1973a) Determination of cellulase activity using hydroxyethylcellulose as substrate. Canadian Journal of Biochemistry 51:39-43.
21. Child, JJ; Knapp, C; Eveleigh, DE. (1973b) Improved pH control of fungal culture media. Mycologia 65:1078-1086.
22. Cronin, LA; Tiffney Jr, WN; Eveleigh, DE. (2000) The graying of cedar shingles in a maritime climate—a fungal basis? Journal of Industrial Microbiology and Biotechnology 24:319-322.
23. Dally, EL; Stokes, HW; Eveleigh, DE. (1982) A genetic comparison of strains of Zymomonas mobilis by analysis of plasmid DNA. Biotechnology Letters 4:91-96.
24. Danmek, K; Prasongsuk, S; Lotrakul, P; Damann, KE; Eveleigh, DE; Punnapayak, H. (2011) Effect of Avid on the synnema-like formation of Aspergillus flavus grown on Czapek medium. African Journal of Microbiology Research 5:2812-2815.
25. Davis, B; Eveleigh, DE. (1984) Chitosanases: occurrence, production and immobilization, p. 161–179. In Zikakis, J (ed.), Chitin, Chitosan, and Related Enzymes, Elsevier, Amsterdam.
26. el-Gogary, S; Leite, A; Crivellaro, O; Eveleigh, DE; el-Dorry, H. (1989) Mechanism by which cellulose triggers cellobiohydrolase I gene expression in Trichoderma reesei. Proceedings of the Natlonal Academy of Science U S A 86:6138-6141.
27. Elliston, KO; Yablonsky, MD; Eveleigh, DE. (1991) Cellulase: insights through recombinant DNA approaches, p. 290–300. In (eds.), Enzymes in Biomass Conversion, American Chemical Society, Washington, DC.
28. Eveleigh, D; Davis, D. (1996) Whimsical wrinkles with Winogradsky’s wonder. SGM Quarterly 23:106-107.
29. Eveleigh, DE; Bok, JD; El-Dorry, H; El-Gogary, S; Elliston, K; Goyal, A; Waldron, C; Wright, R; Wu, YM. (1995) Cellulase lessons revealed through the microbe’s perspective. Applied Biochemistry and Biotechnology 51:169-177.
30. Eveleigh, DE; Stokes, HW; Dally, EL. (1983) Recombinant DNA approaches for enhancing the ethanol productivity of Zymomonas mobilis, p. 69–91. In Wise, DL (ed.), Organic Chemicals from Biomass, Benjamin-Cummings, San Francisco.
31. Eveleigh, DE. (1961) The growth requirements of Phoma violacea, with reference to its disfiguration of painted surfaces. Annals of Applied Biology 49:412-423.
32. Eveleigh, DE. (1983) The fermentation of biomass — current aspects, p. 365–391. In Côté, WA (ed.), Biomass Utilization, Vol. 67,
33. Eveleigh, DE. (1987a) Cellulase: a prespective. Philosophical Transactions of the Royal Society London A 321:435-447.
34. Eveleigh, DE. (1987b) The natural history of the degradation of wood: discussion. Philosophical Transactions of the Royal Society of London. Series B 321:433.
35. Eveleigh, DE. (1992) Nirogen Fixing Organisms (Book Review). Soil Science 153:417.
36. Eveleigh, DE. (2003) Cellulolysis canards - misconceptions. ASM News 69:161-161.
37. Eveleigh, DE. (1985) Trichoderma, p. In Demain, AL; Solomon, NA (eds.), Biology of Industrial Microorganisms, Butterworth-Heinemann, Oxford.
38. Eveleigh, DE. (1961a) Phoma spp. associated with painted surfaces. Transactions of the British Mycological Society 44:573-585.
39. Eveleigh, DE. (1961b) The disfiguration of painted surfaces by fungi, with special reference to Phoma violacea. Annals of Applied Biology 49:403-411.
40. Eveleigh, DE. (1967) The determination of β-1, 3-glucanase: anomalous reduction of alkaline 3,5-dinitrosalicylate. Canadian Journal of Microbiology 13:727-729.
41. Eveleigh, DE. (1970) Fungal disfigurement of paper, and soft rot of cedar shingles. Applied Microbiology 19:872-874.
42. Eveleigh, DE. (1979) Soil Enzymes (Book Review). Soil Science 128:191.
43. Eveleigh, DE. (2012) Mushrooms for the millions: review of: Mycophilia—Revelations from the Weird World of Mushrooms. Journal of Microbiology & Biology Education 13:103-104.
44. Eveleigh, DE; Bennett, JW. (2013) Gerhard Haas (1917-2013). Microbe 8:423-424.
45. Eveleigh, DE; Brewer, D. (1963) Studies on slime accumulations in pulp and paper mills: VI. Isolation of thermophilic and thermotolerant fungi from paper mills. Canadian Journal of Botany 41:1377-1382.
46. Eveleigh, DE; Brewer, D. (1964a) Ecological observations on the fungi and bacteria in slime accumulations in a paper mill. Canadian Journal of Botany 42:35-43.
47. Eveleigh, DE; Brewer, D. (1964b) Nutritional requirements of the microflora of a slime accumulation in a paper mill. Canadian Journal of Botany 42:341-350.
48. Eveleigh, DE; Brewer, D. (1965) Interrelationships between microorganisms in an accumulation of slime in a paper mill. Canadian Journal of Botany 43:519-526.
49. Eveleigh, DE; Chase, T. (1997) Presidential discovery that methane is a gas. ASM News 63:63-63.
50. Eveleigh, DE; Child, JJ. (1969) Use of non-ionic substrates for determination of cellulase. Fungal Genetics Reports 14:28.
51. Eveleigh, DE; Häggblom, M. (2013) The Rutgers antibiotics. Chemical & Engineering News 91:2.
52. Eveleigh, DE; Häggblom, M; Boyd, J. (2015) The Early Challenges of Antibiotic Discovery. Microbe 10:449-450.
53. Eveleigh, DE; Mandels, M; Andreotti, R; Roche, C. (2009) Measurement of saccharifying cellulase. Biotechnology for Biofuels 2:21.
54. Eveleigh, DE; Perlin, AS. (1969) A proton magnetic resonance study of the anomeric species produced by D-glucosidases. Carbohydrate Research 10:87-95.
55. Eveleigh, DE; Sietsma, JH; Haskins, RH. (1968) The involvement of cellulase and laminaranase in the formation of Pythium protoplasts. Microbiology 52:89-97.
56. Eveleigh, DE; Dateo, GP; Reese, ET. (1964) Fungal metabolism of complex glycosides: ustilagic acid. Journal of Biological Chemistry 239:839-844.
57. Eveleigh, DE; Montenecourt, BS. (1979) Increasing yields of extracellular enzymes, p. 57–74. In Perlman, D (ed.), Advances in Applied Microbiology, Vol. 25, Elsevier, Amsterdam.
58. Eveleigh, DE. (1981) The microbiological production of industrial chemicals. Scientific American 245:154-179.
59. Eveleigh, DE. (1994) Fungal enzymes. Mycologist 8:44.
60. Eveleigh, DE; Bennett, JW. (2018) Women microbiologists at Rutgers in the early golden age of antibiotics, p. 317–329. In Whitaker, RJ; Barton, HA (eds.), Women in Microbiology, ASM Press, Washington, DC.
61. Eveleigh, DE; Waldron, CR; Bartley, T. (1986) Method for the conversion of a cellulosic substrate to glucose using Microbispora bispora, strain Rutgers P&W. Patent # 4,628,029.
62. Fenton, DM; Eveleigh, DE. (1981) Purification and mode of action of a chitosanase from Penicillium islandicum. Microbiology 126:151-165.
63. Fenton, DM; Davis, B; Rotgers, C; Eveleigh, DE. (1978) Enzymatic hydrolysis of chitosan, p. 525–541. In Muzzarrelli, RAA; Pariser, ER (eds.), First International Symposium on Chitin and Chitosan, MIT Press, Boston.
64. Flavell, RB; Barz, W; Peacock, WJ; Beringer, JE; Saedler, H; Broda, P; Salamini, F; Eveleigh, DE; Starlinger, P; Hahlbrock, K; Weiler, E; Kahmann, R; Zenk, M. (1985) Plants and agriculture, p. 199–222. In Silver, S (ed.), Biotechnology: Potentials and Limitations, Springer-Verlag, Berlin.
65. Frein, EM; Montenecourt, BS; Eveleigh, DE. (1982) Cellulase production by Trichoderma reesei immobilized on κ-carrageenan. Biotechnology Letters 4:287-292.
66. Gamborg, OL; Eveleigh, DE. (1968) Culture methods and detection of glucanases in suspension cultures of wheat and barley. Canadian Journal of Biochemistry 46:417-421.
67. Ghosh, A; Al-Rabiai, S; Ghosh, BK; Trimiño-Vazquez, H; Eveleigh, DE; Montenecourt, BS. (1982) Increased endoplasmic reticulum content of a mutant of Trichoderma reesei (RUT-C30) in relation to cellulase synthesis. Enzyme and Microbial Technology 4:110-113.
68. Ghosh, A; Ghosh, BK; Trimino-Vazquez, H; Eveleigh, DE; Montenecourt, BS. (1984) Cellulase secretion from a hyper-cellulolytic mutant of Trichoderma reesei Rut-C30. Archives of Microbiology 140:126-133.
69. Gorin, PAJ; Spencer, JFT; Eveleigh, DE. (1969) Enzymic degradation of yeast cell-wall mannans and galactomannans to polymeric fragments containing α-(1→ 6)-linked D-mannopyranose residues. Carbohydrate Research 11:387-398.
70. Gorin, PAJ; Eveleigh, DE. (1970) Extracellular 2-acetamido-2-deoxy-D-galacto-D-galactan from Aspergillus nidulans. Biochemistry 9:5023-5027.
71. Gottlund, K; Eveleigh, DE. (1988) Bland Symington Montenecourt, 1942–1987. Mycologia 80:767-768.
72. Goyal, AK; Eveleigh, DE. (1996) Cloning, sequencing and analysis of the ggh-A gene encoding a 1,4-beta-D-glucan glucohydrolase from Microbispora bispora. Gene 172:93-98.
73. Goyal, A; Eveleigh, DE. (1992) Degradation of cellulose and effects of metal ions on cellulases, p. 399–414. In Sigel, H; Sigel, A (eds.), Metal Ions in Biological Systems, CRC Press, Boca Raton, FL.
74. Goyal, A; Ghosh, B; Eveleigh, D. (1991) Characteristics of fungal cellulases. Bioresource Technology 36:37-50.
75. Häggblom, M; Eveleigh, DE; Boyd, J; Warhol, J. (2019) Mining for microbes. Chemical & Engineering News 97:4-5.
76. Hu, P; Chase, T; Eveleigh, DE. (1993) Cloning of a Microbispora bispora cellobiohydrolase gene in Streptomyces lividans. Applied Microbiology and Biotechnology 38:631-637.
77. Hu, P; Kahrs, SK; Chase, T; Eveleigh, DE. (1992) Cloning of a Microbispora bispora cellobiohydrolase gene in Escherichia coli. Journal of Industrial Microbiology 10:103-110.
78. Hunter-Cevera, JC; Eveleigh, DE. (1990) Actinomycetes, p. 33–47. In Dindal, DL (ed.), Soil Biology Guide, Wiley, New York.
79. Hunter-Cevera, JC; Jeffries, TW; Eveleigh, DE. (1996) Introduction. Journal of Industrial Microbiology 17:vi.
80. Jeffries, TW; Eveleigh, DE; Macmillan, JD; Parrish, FW; Reese, ET. (1977) Enzymatic hydrolysis of the walls of yeasts cells and germinated fungal spores. Biochimica et Biophysica Acta (BBA)-General Subjects 499:10-23.
81. Kelleher, TJ; Montenecourt, BS; Eveleigh, DE. (1987) Cellobiose-quinone oxidoreductase —application in monitoring cellobiohydrolase purification. Applied Microbiology and Biotechnology 27:299-305.
82. Keller, WA; Harvey, B; Gamborg, OL; Miller, RA; Eveleigh, DE. (1970) Plant protoplasts for use in somatic cell hybridization. Nature 226:280.
83. King, MR; Yernool, DA; Eveleigh, DE; Chassy, BM. (1998) Thermostable alpha-galactosidase from Thermotoga neapolitana: cloning, sequencing and expression. FEMS Microbiology Letters 163:37-42.
84. Klein, D; Eveleigh, DE. (1998) Ecology of Trichoderma, p. 57–74. In Harman, GE; Kubicek, CP (eds.), Trichoderma and Gliocladium Vol. 1, CRC Press, London.
85. Kubicek, CP; Eveleigh, DE; Esterbauer, H; Steiner, W; Kubicek-Pranz, EM. (1990) Trichoderma Cellulases: Biochemistry, Physiology, Genetics and Applications, (eds.), The Royal Society Chemistry, Cambridge.
86. Lam, CK; O’Mullan, P; Eveleigh, DE. (1993) Transformation of Zymononas mobilis by electroporation. Applied Microbiology and Biotechnology 39:305-308.
87. Lejeune, A; Colson, C; Eveleigh, DE. (1986) Cloning of an endoglucanase gene from Pseudomonas fluorescens var. cellulosa into Escherichia coli and Pseudomonas fluorescens. Journal of Industrial Microbiology & Biotechnology 1:79-86.
88. Lejeune, A; Eveleigh, DE; Colson, C. (1988) Expression of an endoglucanase gene of Pseudomonas fluorescens var. cellulosa in Zymomonas mobilis. FEMS Microbiology Letters 49:363-366.
89. Lima, AO; Davis, DF; Swiatek, G; McCarthy, JK; Yernool, D; Pizzirani-Kleiner, AA; Eveleigh, DE. (2009) Evaluation of GFP tag as a screening reporter in directed evolution of a hyperthermophilic beta-glucosidase. Molecular Biotechnology 42:205-215.
90. Linke, HAB; Eveleigh, DE. (1975a) Facile synthesis of the chalcones and dihydrochalcones from naringin, neohesperidin and hesperidin. Zeitschrift für Naturforschung B 30:606-608.
91. Linke, HAB; Eveleigh, DE. (1975b) Dihydrochalcones and chalcones of some flavanone glycosides: qualitative and quantitative determinations. Zeitschrift für Naturforschung B 30:940-942.
92. Linke, HAB; Eveleigh, DE. (1978) Sweetening foods with neohesperidin chalcone. USA Patent # 4,087,558.
93. Mandels, M; Eveleigh, DE. (2009) Reflections on the United States military 1941-1987. Biotechnology for Biofuels 2:20.
94. Manitchotpisit, P; Leathers, TD; Peterson, SW; Kurtzman, CP; Li, XL; Eveleigh, DE; Lotrakul, P; Prasongsuk, S; Dunlap, CA; Vermillion, KE; Punnapayak, H. (2009) Multilocus phylogenetic analyses, pullulan production and xylanase activity of tropical isolates of Aureobasidium pullulans. Mycological Research 113:1107-1120.
95. Manitchotpisit, P; Skory, CD; Leathers, TD; Lotrakul, P; Eveleigh, DE; Prasongsuk, S; Punnapayak, H. (2011) Alpha-amylase activity during pullulan production and alpha-amylase gene analyses of Aureobasidium pullulans. Journal of Industrial Microbiology and Biotechnology 38:1211-1218.
96. McCarthy, JK; O’Brien, CE; Eveleigh, DE. (2003) Thermostable continuous coupled assay for measuring glucose using glucokinase and glucose-6-phosphate dehydrogenase from the marine hyperthermophile Thermotoga maritima. Analytical Biochemistry 318:196-203.
97. McCarthy, JK; Uzelac, A; Davis, DF; Eveleigh, DE. (2004) Improved catalytic efficiency and active site modification of 1,4-beta-D-glucan glucohydrolase A from Thermotoga neapolitana by directed evolution. Journal of Biological Chemistry 279:11495-11502.
98. Monaghan, RL; Eveleigh, DE; Tewari, RP; Reese, ET. (1973) Chitosanase, a novel enzyme. Nature New Biology 245:78-80.
99. Montenecourt, BS; Eveleigh, DE. (1979) Selective screening methods for the isolation of high yielding cellulase mutants of Trichoderma reesei, p. 289–301. In Brown, RD; Jurasek, L (eds.), Advances in Chemistry, Volume 181, AMERICAN CHEMICAL SOCIETY,
100. Montenecourt, BS; Eveleigh, DE. (1977a) Semiquantitative plate assay for determination of cellulase production by Trichoderma viride. Applied and Environmental Microbiology 33:178-183.
101. Montenecourt, BS; Eveleigh, DE; Elmund, GD; Parcells, J. (1978) Antibiotic disks—an improvement in the filter paper assay for cellulase. Biotechnology and Bioengineering 20:297-300.
102. Montenecourt, BS; Eveleigh, DE. (1977b) Preparation of mutants of Trichoderma reesei with enhanced cellulase production. Applied and Environmental Microbiology 34:777-782.
103. Montenecourt, BS; Nhlapo, SD; Trimino-Vazquez, H; Cuskey, S; Schamhart, DH; Eveleigh, DE. (1981) Regulatory controls in relation to over-production of fungal cellulases. Basic Life Science 18:33-53.
104. Morris, M; Eveleigh, DE; Riggs, SC; Tiffney Jr, WN. (1974) Nitrogen fixation in the bayberry (Myrica pensylvanica) and its role in coastal succession. American Journal of Botany 61:867-870.
105. (1992) Microbial Metabolites, Nash, C; Hunter-Cevera, J; Cooper, R; Eveleigh, DE; Hamill, R (eds.), Wm. C. Brown, Dubuque.
106. Natarajan, S; Eveleigh, DE; Dawson, RH. (1976) A natural source for Neohesperidin. Economic Botany 30:38-38.
107. Nimchua, T; Eveleigh, DE; Sangwatanaroj, U; Punnapayak, H. (2008) Screening of tropical fungi producing polyethylene terephthalate-hydrolyzing enzyme for fabric modification. Journal of Industrial Microbiology and Biotechnology 35:843-850.
108. O’Mullan, P; Szakacs-Dobozi, M; Eveleigh, DE. (1991) Identification of saccharolytic enzymes of Zymomonas mobilis CP4. Biotechnology Letters 13:137-142.
109. O’Mullan, PJ; Chase, T; Eveleigh, DE. (1992) Purification and some properties of extracellular invertase B from Zymomonas mobilis. Applied Microbiology and Biotechnology 38:341-346.
110. O’Mullan, PJ; Buchholz, SE; Chase, T; Eveleigh, DE. (1995) Roles of alcohol dehydrogenases of Zymomonas mobilis (ZADH): characterization of a ZADH-2-negative mutant. Applied Microbiology and Biotechnology 43:675-678.
111. Permpornsakul, P; Prasongsuk, S; Lotrakul, P; Eveleigh, DE; Kobayashi, DY; Imai, T; Punnapayak, H. (2016) Biological treatment of reactive black 5 by resupinate white rot fungus Phanerochaete sordida PBU 0057. Polish Journal of Environmental Studies 25:1167.
112. Permpornsakula, P; Prasongsuka, S; Lotrakula, P; Eveleigh, DE; Kobayashi, DY; Wud, SH; Imaie, T; Punnapayaka, H. (2016) Two new records of the resupinate polypore fungi, Ceriporia cystidiata and Macrohyporia dictyopora, in Thailand. ScienceAsia 42:171-177.
113. Picataggio, SK; Schamhart, DHJ; Montenecourt, BS; Eveleigh, DE. (1983) Sphaeroplast formation and regeneration in Trichoderma reesei. European Journal of Applied Microbiology and Biotechnology 17:121-128.
114. Prasongsuk, S; Berhow, MA; Dunlap, CA; Weisleder, D; Leathers, TD; Eveleigh, DE; Punnapayak, H. (2007) Pullulan production by tropical isolates of Aureobasidium pullulans. Journal of Industrial Microbiology and Biotechnology 34:55-61.
115. Prasongsuk, S; Sullivan, RF; Kuhirun, M; Eveleigh, DE; Punnapayak, H. (2005) Thailand habitats as sources of pullulan-producing strains of Aureobasidium pullulans. World Journal of Microbiology and Biotechnology 21:393-398.
116. Riske, F; Labudova, I; Miller, L; Macmillan, JD; Eveleigh, DE. (1987) Monoclonal antibodies against cellobiohydrolase from Trichoderma reesei, p. 167–176. In Moo-Young, M (ed.), Biomass Conversion Technology, Pergamon, New York.
117. Riske, FJ; Eveleigh, DE; Macmillan, JD. (1986) Purification of cellobiohydrolase I from Trichoderma reesei using monoclonal antibodies in an immunomatrix. Journal of Industrial Microbiology 1:259-264.
118. Riske, FJ; Eveleigh, DE; Macmillan, JD. (1990) Double-antibody sandwich enzyme-linked immunosorbent assay for cellobiohydrolase I. Applied and Environmental Microbiology 56:3261-3265.
119. Ritacco, FV; Eveleigh, DE. (2008) Molecular and phenotypic comparison of phaeochromycin-producing strains of Streptomyces phaeochromogenes and Streptomyces ederensis. Journal of Industrial Microbiology and Biotechnology 35:931-945.
120. Saraihom, S; Kobayashi, DY; Lotrakul, P; Prasongsuk, S; Eveleigh, DE; Punnapayak, H. (2016) First report of a tropical Lysobacter enzymogenes producing bifunctional endoglucanase activity towards carboxymethylcellulose and chitosan. Annals of Microbiology 66:907-919.
121. Schimenti, J; Garrett, T; Montenecourt, BS; Eveleigh, DE. (1983) Selection of hypercellulolytic mutants of Trichoderma reesei based on resistance to nystatir. Mycologia 75:876-880.
122. Sietsma, JH; Eveleigh, DE; Haskins, RH. (1968) The purification of cellulase and exo-laminaranase and their role in the formation of Pythium sp.“protoplasts”. Antonie van Leeuwenhoek 34:331-340.
123. Sietsma, JH; Eveleigh, DE; Haskins, RH. (1969) Cell wall composition and protoplast formation of some Oomycete species. Biochimica et Biophysica Acta 184:306-317.
124. Sietsma, JH; Eveleigh, DE; Haskins, RH; Spencer, JFT. (1967) “ Protoplasts” from Pythium sp. PRL 2142. Canadian Journal of Microbiology 13:1701-1704.
125. Stokes, HW; Picataggio, SK; Eveleigh, DE. (1983a) Recombinant genetic approaches for efficient ethanol production, p. 113–132. In Böer, KW; Duffie, JA (eds.), Advances in Solar Energy, Springer, Boston, MA.
126. Stokes, HW; Dally, EL; Yablonsky, MD; Eveleigh, DE. (1983b) Comparison of plasmids in strains of Zymomonas mobilis. Plasmid 9:138-146.
127. Tiffney Jr, WN; Benson, DR; Eveleigh, DE. (1978) Does Arctostaphylos uva‐ursi (bearberry) have nitrogen‐fixing root nodules? American Journal of Botany 65:625-628.
128. Waldron, CR; Becker-Vallone, CA; Eveleigh, DE. (1986) Isolation and characterization of a cellulolytic Actinomycete Microbispora bispora. Applied Microbiology and Biotechnology 24:477-486.
129. Waldron, CR; Eveleigh, DE. (1986) Saccharification of cellulosics by Microbispora bispora. Applied Microbiology and Biotechnology 24:487-492.
130. Waldron, CRJ; Frein, EM; Eveleigh, DE. (1983) Prevention of phosphate interference in HPLC sugar analysis studies. Biotechnology and Bioengineering 25:2091-2092.
131. Wright, RM; Yablonsky, MD; Shalita, ZP; Goyal, AK; Eveleigh, DE. (1992) Cloning, characterization, and nucleotide sequence of a gene encoding Microbispora bispora BglB, a thermostable beta-glucosidase expressed in Escherichia coli. Applied and Environmental Microbiology 58:3455-3465.
132. Yablonsky, MD; Bartley, T; Elliston, KO; Kahrs, SK; Shalita, ZP; Eveleigh, DE. (1988a) Characterization and cloning of the cellulase complex of Microbispora bispora, p. 249–266. In Aubert, JP; Béguin, P; Millet, J (eds.), Biochemistry and Genetics of Cellulose Degradation, Academic, London.
133. Yablonsky, MD; Goodman, AE; Stevnsborg, N; Gonçalves de Lima, O; Falcão de Morais, JO; Lawford, HG; Rogers, PL; Eveleigh, DE. (1988b) Zymomonas mobilis CP4: a clarification of strains via plasmid profiles. Journal of Biotechnology 9:71-79.
134. Yablonsky, MD; Elliston, KO; Eveleigh, DE. (1989) The relationship between the endoglucanase MbcelA of Microbispora bispora and the cellulases of Cellulomonas fimi, p. 73–83. In (eds.), Enzyme Systems for Lignocellulose Degradation, Elsevier Applied Sciences, London.
135. Yanwisetpakdee, B; Lotrakul, P; Prasongsuk, S; Seelanan, T; White Jr, JF; Eveleigh, DE; Kim, SW; Punnapayak, H. (2016) Associations among halotolerance, osmotolerance and exopolysaccharide production of Aureobasidium melanogenum strains from habitats under salt stress. Pakistan Journal of Botany 48:1229-1239.
136. Yernool, DA; McCarthy, JK; Eveleigh, DE; Bok, JD. (2000a) Cloning and characterization of the glucooligosaccharide catabolic pathway beta-glucan glucohydrolase and cellobiose phosphorylase in the marine hyperthermophile Thermotoga neapolitana. Journal of Bacteriology 182:5172-5179.
137. Yernool, D; Eveleigh, DE; King, MR; Chassy, B. (2000b) Thermostable alpha-galactosidase and methods of use. USA Patent # 6,150,171.
138. Zitomer, SW; Eveleigh, DE. (1987) Cellulase screening by iodine staining: an artefact. Enzyme and Microbial Technology 9:214-216.
.












Comments